The global market for rheumatoid arthritis treatments is expected to grow at a CAGR of...
Learn More
Our consulting solutions address company specific challenges with respect to micro environment...
Learn More
Organizations frequently need day-today research guidancein order to gain strategic...
Learn More
Exploring different areas of market research and market analysis is a key factor...
Learn MoreAcute Market Reports presents the most extensive global business research services across industries. Our research studies focus on potential outcomes, benefits, and risks associated with each market segment across geographies. Having served our global clients for more than 10 years, our prime priority is to enable our clients in making well-informed business decisions through a data-driven, analytical, and uncomplicated research approach.
We provide access to the world's most comprehensive, analytical, and updated business intelligence services and solutions.



The diesel generator market in the telecom industry has shown significant dynamics over recent years. With the rapid expansion of the telecom sector, the demand for reliable power solutions has grown exponentially. Diesel generators have been a primary source of backup and emergency power for tel...
Read More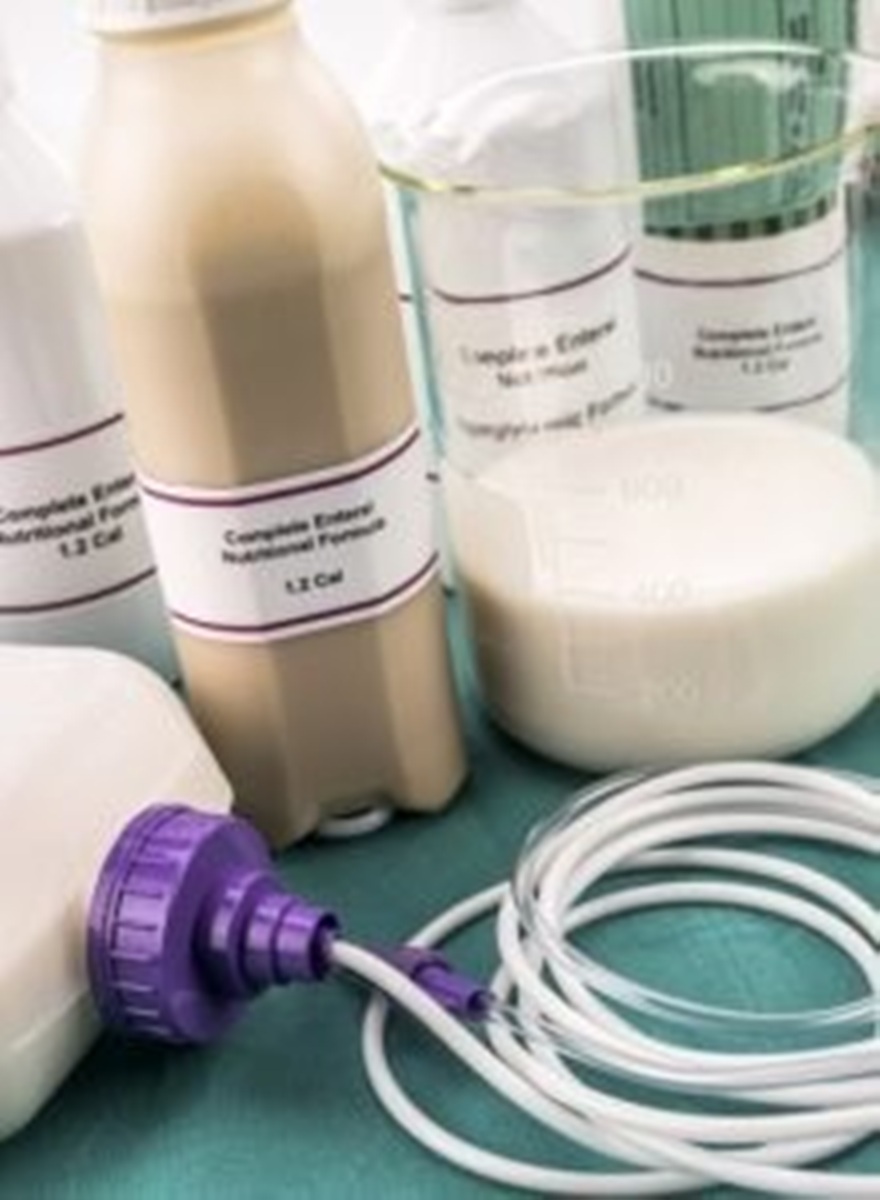
The enteral feeding formulas market encompasses a range of nutritional solutions administered to patients via the gastrointestinal tract when oral intake is inadequate, impossible, or unsafe. This method of feeding is used across various healthcare settings including ...
Read More
The wearable camera market is expected to witness a CAGR of 14.5% during the forecast period of 2025 to 2033, driven by several key factors. The wearable camera market has evolved to meet the demands of a tech-savvy generation. Wearable cameras, ofte...
Read More